Disruption of PI3K/OxPhos Coupling by Trehalose Drives a BCAA to Lipid Metabolic Switch in Hormone Receptor Positive Breast Cancer
Disruption of PI3K/OxPhos Coupling by Trehalose Drives a BCAA to Lipid Metabolic Switch in Hormone Receptor Positive Breast Cancer
Singh, T.; Kaur, P.; Munshi, A.; Singh, S.
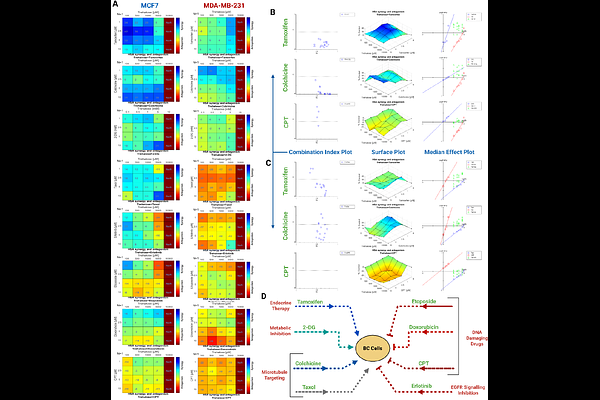
AbstractCancer cells sustain proliferation through dynamic coordination between mitochondrial oxidative phosphorylation (OxPhos) and anabolic carbon metabolism. How this metabolic coupling can be selectively destabilized in subtype-specific contexts remains poorly defined. Here we identify trehalose, a disaccharide previously linked to autophagy modulation, as a regulator of mitochondrial anabolic integration in breast cancer. Using high-resolution respirometry, untargeted metabolomics, and signalling analyses across estrogen/progesterone receptor positive and triple negative breast cancer models, we show that trehalose preferentially impairs mitochondrial bioenergetics in OxPhos-dependent ER positive cells. Trehalose reduced electron transport system capacity, NADH linked respiration, mitochondrial membrane potential, and coupling efficiency, while suppressing mitochondrial biogenesis markers. These bioenergetic effects coincided with attenuation of PI3K/Akt signalling and induction of p21-associated growth arrest. Metabolomic profiling revealed a coordinated redistribution of carbon flux characterized by depletion of branched-chain amino acids (BCAAs) and glycolytic intermediates alongside accumulation of long-chain fatty acids and cholesterol. Correlation network analysis uncovered a strong inverse relationship between BCAA linked metabolism and lipid abundance, indicating a regulated metabolic trade-off rather than nonspecific stress. Functionally, trehalose enhanced the efficacy of mitochondrial interfering agents such as tamoxifen and colchicine, while exerting minimal effects in metabolically flexible TNBC cells. Together, these findings define trehalose as a metabolic modulator that constrains mitochondrial plasticity and enforces a lipid-buffered, growth-restrictive state in ER positive breast cancer, revealing a therapeutic vulnerability linked to mitochondrial dependency.