Chromatin association promotes UBR5-mediated degradation of Rb
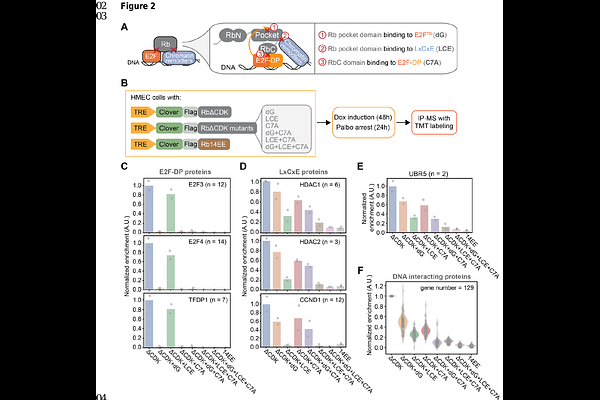
Chromatin association promotes UBR5-mediated degradation of Rb
Zhang, S.; Lanz, M.; Konschnik, J.; Skotheim, J.
AbstractThe retinoblastoma protein Rb is a cell cycle inhibitor that plays a central role in regulating the G1/S cell cycle transition. Un-/hypo-phosphorylated Rb suppresses E2F transcription activity by binding to E2F/DP dimers and recruiting chromatin remodelers to prevent cells from entering S phase. For cells to progress through the G1/S transition, Rb is inactivated by two mechanisms: the classic pathway of Rb hyperphosphorylation by Cyclin-CDK complexes, and a recently identified degradation mechanism driven by the E3 ubiquitin ligase UBR5. These two pathways are interconnected, as only the un-/hypo-phosphorylated Rb can be degraded, and the hyper-phosphorylated Rb is stabilized to promote its reaccumulation in preparation for the next cell division cycle. However, the molecular basis for how Rb is stabilized upon phosphorylation remains unclear. In this study, we found that UBR5 preferentially targets chromatin-associated proteins for degradation. Since the chromatin association of Rb is modulated by its phosphorylation, we hypothesized that phosphorylation may affect Rb stability by altering its chromatin association. To test this, we constructed a series of un-phosphorylatable Rb variants with graded reductions in chromatin association. Consistent with our hypothesis, we observed a strong correlation between the chromatin association of a Rb variant and its half-life. Fusing these Rb variants to histone H1 increased chromatin association to similar levels and equalized their protein half-lives. Taken together, these findings show how phosphorylation stabilizes Rb by promoting its dissociation from chromatin. This provides a striking example for how sub-organellar protein localization may be used to regulate stability.