A cinnamyl alcohol dehydrogenase scaffold organizes monoterpenoid indole alkaloid biosynthesis
A cinnamyl alcohol dehydrogenase scaffold organizes monoterpenoid indole alkaloid biosynthesis
Gao, D.; Mann, S. G. A.; Chen, B.; Gou, Y.; Chen, C.; Garza-Garcia, J. J. O.; Shahsavarani, M.; Jiang, X.; Tran, H. C.; Bao, J.; Richardson, M. B.; Li, J.; Perley, J. O.; Hwang, J.; Dong, F.; Dong, C.; Huang, L.; De Luca, V.; Wang, Y.; Qu, Y.; Lian, J.
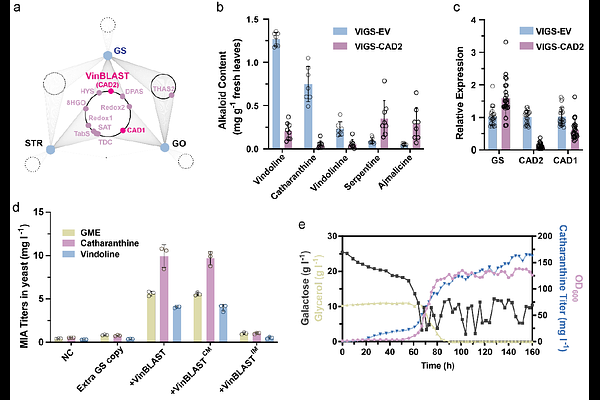
AbstractBiosynthesis of ~3000 monoterpenoid indole alkaloids (MIAs) including the anticancer drug vinblastine involve the highly unstable intermediate strictosidine aglycone. Its formation by strictosidine {beta}-glucosidase (SGD) and subsequent conversion by geissoschizine synthase (GS) occur in spatially separated compartments, representing a major biosynthesis bottleneck. Here we discover VinBLAST, a cinnamyl alcohol dehydrogenase repurposed as a scaffold for efficient processing of this labile intermediate. VinBLAST physically mediates SGD and GS interaction in the nucleus and allosterically enhances GS catalytic efficiency. VinBLAST homologs from diverse plant families enhance biosynthesis of several representative MIAs, with the production of catharanthine increased to ~160 mg l-1 in yeast, nearly 1000-fold higher than previous studies. Our discovery provides the missing link in organizing MIA biosynthesis and enables scalable bioproduction of geissoschizine-derived therapeutics.