Molecular mechanisms of PDCD4-mediated modulation of translation initiation and termination
Molecular mechanisms of PDCD4-mediated modulation of translation initiation and termination
Al Sheikh, W.; Shuvalova, E.; Biziaev, N.; Salman, A.; Kolosov, P.; Shuvalov, A.; Alkalaeva, E.
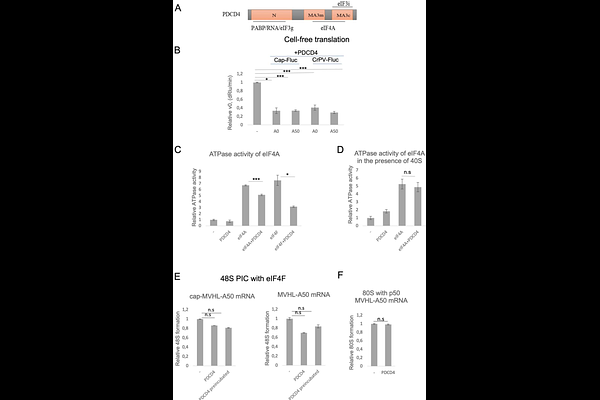
AbstractPDCD4 is a tumor suppressor, known to affect protein translation by binding to a component of the eIF4F complex, eIF4A, and reducing its helicase activity, which is necessary for the 48S preinitiation complex formation and scanning of the 5' untranslated region of mRNA. PDCD4 has also been shown to interact with the ribosome and with translation initiation factors eIF4G, eIF4G2, eIF3, and PABP, all of which participate both in initiation and the closed-loop structure that couples initiation and termination. To investigate whether PDCD4 modulates initiation and termination through these interactions, we used a reconstituted mammalian translation system and pre-termination complexes purified from rabbit reticulocyte lysate. We found that PDCD4 suppresses early initiation events prior to eIF4F complex binding to the cap structure on mRNA. Moreover, inhibition of the helicase activity of eIF4A by PDCD4 is lost when the 40S subunit is present. Inhibition of 48S complex formation was also observed in the presence of the truncated eIF4G fragment p50 or the eIF4G2 isoform, both of which interact with eIF4A but lack the eIF4E-binding domain. PDCD4-mediated inhibition of initiation persisted regardless of the presence of PABP. During translation termination, PDCD4 did not affect eIF4A activity, indicating that its regulatory function toward eIF4A is stage-specific and restricted to initiation. Finally, we discovered that PDCD4 additively stimulates peptide release together with eIF3, eIF4G2, and PABP, but competes with eIF4F. Thus, PDCD4 employs a complex molecular mechanism targeting multiple translation factors to regulate different stages of protein synthesis.