Targeting CLK1 by CRISPR tools and pharmacological inhibition modulates the innate immune response and dengue virus replication
Targeting CLK1 by CRISPR tools and pharmacological inhibition modulates the innate immune response and dengue virus replication
Pozzi, B.; Lucero, A. M.; SREBROW, A.
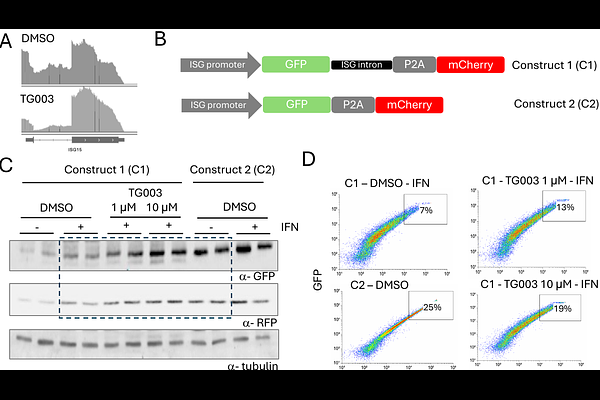
AbstractAlternative splicing is a key regulatory mechanism known to be altered upon viral infection. These alterations can arise both from direct viral interference with the splicing machinery and from cellular responses such as activation of innate immunity. Here, we investigated splicing changes shared between dengue virus (DENV) infection and interferon (IFN) treatment in cultured cells, the latter serving as a model for virus-independent innate immune activation. Among the common events, we identified an increased production of non-coding mRNA isoforms from the CLK1 gene, which gives rise to a kinase that phosphorylates splicing factors including SR proteins and spliceosomal components. Consistent with this finding, IFN treatment led to a reduction in CLK1 protein levels. Using stable cell lines with CRISPR/dCas9-mediated modulation of CLK1 expression, we found that silencing CLK1 enhanced the induction of immune response genes, while its over-expression attenuated it. Inhibition of CLK1 kinase activity with the pharmacological inhibitor TG003 further potentiated IFN-induced gene expression and reduced DENV replication. Altogether, these results identify CLK1 as a proviral negative regulator of IFN-stimulated gene expression and suggest that its inhibition could enhance antiviral defenses and become a target for therapeutic strategies.