Mapping kidney trait heritability to individual cells reals disease-specific remodeling of genetic risk architecture
Mapping kidney trait heritability to individual cells reals disease-specific remodeling of genetic risk architecture
Hu, H.
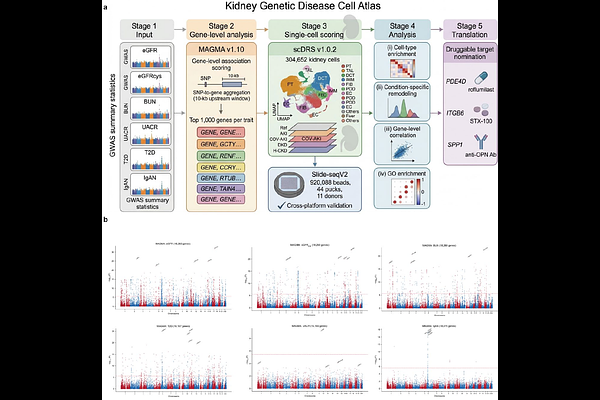
AbstractGenome-wide association studies (GWAS) have identified hundreds of genetic loci associated with kidney function and disease, yet the cell-type-specific mechanisms through which these variants act remain largely unknown. Here, we construct the Kidney Genetic Disease Cell Atlas by applying single-cell disease relevance scoring (scDRS) to map GWAS signals for six kidney-related traits-estimated glomerular filtration rate (eGFR), cystatin C-based eGFR (eGFRcys), blood urea nitrogen (BUN), urinary albumin-to-creatinine ratio (UACR), type 2 diabetes (T2D), and IgA nephropathy (IgAN) onto a comprehensive single-nucleus RNA-seq atlas of 304,652 kidney cells spanning five clinical conditions (healthy reference, acute kidney injury [AKI], COVID-19-associated AKI [COV-AKI], diabetic kidney disease [DKD], and hypertensive chronic kidney disease [H-CKD]). We validate enrichment patterns using Slide-seqV2 spatial transcriptomics from 920,088 beads across 44 pucks, demonstrating strong cross-platform concordance (Spearman rho = 0.72-0.89). Disease-condition-specific analysis reveals dramatic remodeling of genetic risk distribution across cell types, with fibroblasts gaining T2D enrichment in DKD (Delta = +1.07) and immune cells dominating IgAN risk across all conditions (Cohens d = 1.40). Gene-level correlation analysis identifies condition-specific molecular programs, including mitochondrial gene dominance for eGFRcys and PDE4D emergence for T2D/UACR. By integrating scDRS rank shifts with druggability databases, we nominate three high-priority therapeutic targets-PDE4D (roflumilast), ITGB6 (STX-100), and SPP1 (anti-OPN antibody)-each showing disease-specific upregulation in distinct cell populations. The Kidney Genetic Disease Cell Atlas provides a resource for understanding the cellular basis of kidney disease heritability and identifying condition-specific therapeutic opportunities.