Hormone signaling and immune programs define differential endocrine responsiveness in high-risk breast tissue
Hormone signaling and immune programs define differential endocrine responsiveness in high-risk breast tissue
Goldhammer, N.; Bont, M.; Warhadpande, S.; Choi, M.; Cedano, J.; Greenwood, H.; Ye, J.; Schwartz, C.; Alvarado, M.; Ewing, C.; Goodwin, K.; Mukhtar, R.; Wong, J.; Abe, S.; Chandler, J.; Jackson, J.; Olopade, O.; Campbell, M.; Lam, A.; Park, C.; Vertido, A.; van 't Veer, L.; Hylton, N.; Esserman, L.; Rosenbluth, J.
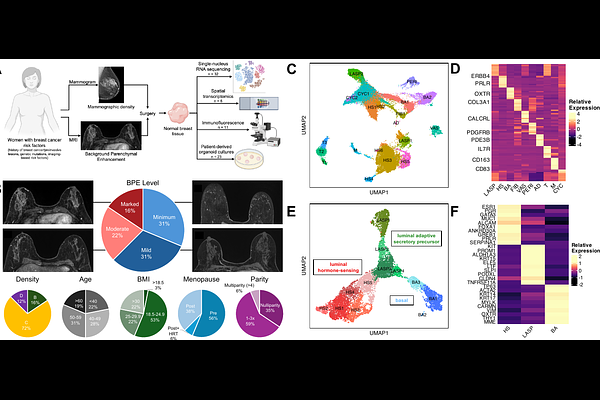
AbstractHormone therapies are frequently used to reduce breast cancer risk in individuals at increased risk for primary or subsequent disease; however, tissue-level responses to these therapies are heterogeneous and incompletely understood. Background parenchymal enhancement (BPE) on breast magnetic resonance imaging (MRI) provides a non-invasive radiologic readout of breast tissue features associated with endocrine responsiveness and cancer risk. Although BPE is associated with hormonal exposure, a subset of patients with BPE do not show a response to preventive endocrine therapy and therefore may remain at increased breast cancer risk. In this study, we integrated single-nucleus RNA sequencing and spatial transcriptomics to define the determinants of endocrine responsiveness in the setting of BPE. We identify hormone-driven epithelial cells with high levels of estrogen signaling and endocrine responsiveness, together with immune-associated epithelial programs characterized by diminished luminal identity and increased expression of immune-modulatory pathways, including major histocompatibility complex (MHC) class II and CD74. Functional organoid assays validate that these epithelial states exhibit differential sensitivity to tamoxifen and demonstrate that inflammatory signals can induce immune-modulatory epithelial programs. Together, our findings identify hormone signaling and immune programs as key determinants of endocrine responsiveness in breast tissue and provide a biological basis for interpreting radiologic markers relevant to cancer prevention.