Slow Dissociation of Nitazenes from the μ-Opioid Receptor Underlies the Challenge of Overdose Reversal
Slow Dissociation of Nitazenes from the μ-Opioid Receptor Underlies the Challenge of Overdose Reversal
Clayton, J.; Kozell, L. B.; Eshleman, A. J.; Bloom, S. H.; Schutzer, W. E.; Abbas, A. I.; Stavitskaya, L.; Shen, J.
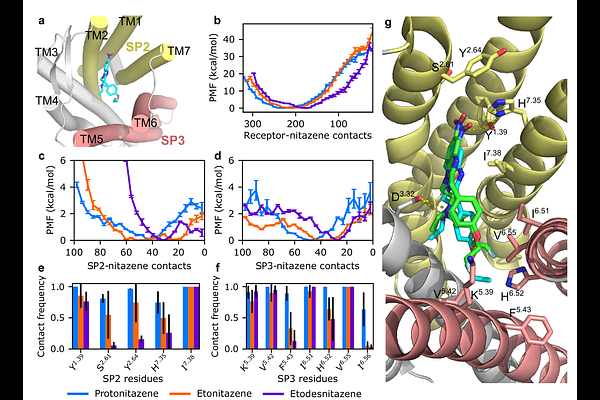
AbstractNitazenes are driving a wave of overdose deaths in the United States and Europe and often require higher doses of naloxone to reverse. To understand the molecular basis, we conducted a joint experimental and simulation study of three common nitazenes, eto-, etodes-, and protonitazene. Radioligand experiments demonstrated that all three nitazenes display higher receptor affinity and longer dissociation half-lives than fentanyl. Notably, protonitazene dissociates slower than carfentanil and its displacement requires fourfold higher antagonist concentrations. The observed trend in nitazene half-lives is recapitulated by molecular dynamics simulations, which suggest that kinetics is controlled by specific interactions with two receptor subpockets. A newly published cryo-EM structure of fluetonitazene-OR complex confirms the predicted interactions, including a -hole bond between the nitro group and Tyr1.39, a residue recently shown to modulate OR signaling bias. Our findings suggest slow receptor dissociation as a key factor challenging overdose reversal. The mechanistic insights have implications for understanding opioid toxicity and designing more effective countermeasures.