Nitric Oxide Modulates Auxin Signaling through TIR1 S-Nitrosylation During Thermomorphogenesis in Arabidopsis
Nitric Oxide Modulates Auxin Signaling through TIR1 S-Nitrosylation During Thermomorphogenesis in Arabidopsis
TEBEZ, N. M.; Correa-Aragunde, N.; Murcia, M. G.; Salvat, S.; Casco, A.; Fiol, D. F.; Casalongue, C. A.; Iglesias, M. J.; Terrile, M. C.
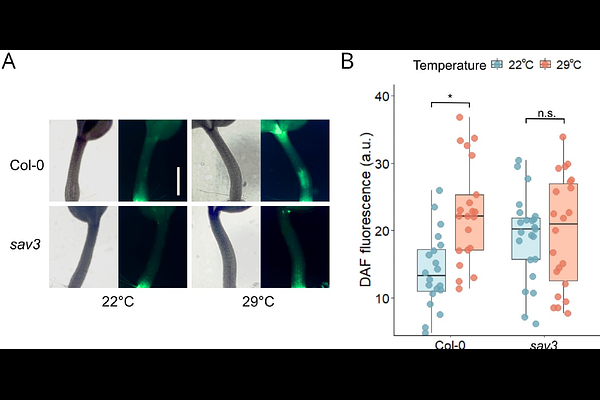
AbstractAuxin, a central hormone coordinating plant growth, integrates both environmental and developmental signals to regulate cell expansion, division, and organ patterning. Among these environmental cues, elevated ambient temperatures trigger a suite of developmental adaptations collectively known as thermomorphogenesis. In this study, we identify nitric oxide (NO) as a key mediator in the temperature-dependent regulation of auxin signaling. Our results show that warm temperatures (28-29 {degrees}C) enhance auxin-induced NO accumulation in Arabidopsis thaliana seedlings. Using pharmacological and genetic approaches, we demonstrate that NO is required for proper thermomorphogenic responses in aerial tissues. This redox signal promotes the stabilization and nuclear localization of the F-box auxin receptor TIR1, a crucial step for the activation of downstream auxin responses. Specifically, tir1-1 seedlings expressing a non-nitrosylatable TIR1 variant mutated at the Cys140 residue exhibit impaired hypocotyl elongation and hyponasty under warm conditions compared to seedlings complemented with wild-type TIR1. These results highlight the functional relevance of the TIR1 Cys140 residue, a known target for S-nitrosylation, in coordinating thermomorphogenic responses. In contrast, the absence of TIR1 S-nitrosylation restricts primary root elongation at 22 {degrees}C but does not affect the growth-promoting effects of warm temperatures. Our findings uncover a novel redox-dependent regulatory layer in auxin signaling, where S-nitrosylation of TIR1 may modulate its stability and subcellular localization in a temperature- and organ-specific manner. This mechanism allows differential growth responses between shoot and root organs and highlights the complexity of hormonal and redox interplay during plant adaptation to elevated temperatures.