The exocyst is an insulin-sensitive regulator of amyloid precursor protein trafficking and amyloid-beta generation in neurons
The exocyst is an insulin-sensitive regulator of amyloid precursor protein trafficking and amyloid-beta generation in neurons
Balaan, C.; Patwardhan, G. Y.; Sachs, R. K.; Kumasaka, H.; Sadagopan, S.; Aou, S.; Lee, A. J.; Nelson, L. T.; Hew, B. E.; Owens, J. B.; Polgar, N.; Ortega, M. A.; Nichols, R. A.; Fogelgren, B.
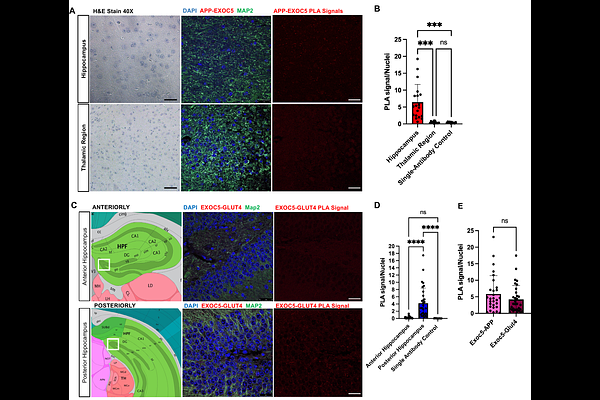
AbstractIntracellular trafficking of amyloid precursor protein (APP) critically influences amyloidogenic processing, yet the mechanisms regulating this pathway remain incompletely defined. The exocyst is a highly conserved, insulin-responsive, eight-subunit tethering complex that directs vesicle targeting and fusion. Based on prior work and preliminary data, we hypothesized that the exocyst plays a central role in neuronal APP trafficking. Using high-resolution imaging, we observed co-localization of APP with multiple exocyst subunits on intracellular vesicles in differentiated human SH-SY5Y neurons and mouse primary hippocampal neurons. These interactions were confirmed by proximity ligation assays demonstrating close APP-Exoc5 association (<40 nm). Following insulin addition, APP-Exoc5 interactions were markedly reduced, whereas, Glut4-Exoc5 interactions significantly increased, indicating insulin-dependent redistribution of exocyst engagement between cargoes. To examine insulin regulation of exocyst-dependent Glut4 trafficking, we generated a pHluorin-Glut4 reporter cell line. Pharmacological inhibition of the exocyst and PI3K signaling reduced Glut4 plasma membrane localization even in the presence of insulin. In SH-SY5Y cells expressing mutant APP that enhances A{beta} production, RNAi-mediated silencing of exocyst subunits significantly decreased sAPP and A{beta} secretion, leading to significant intracellular APP accumulation. Live-cell TIRF microscopy identified exocyst-marked sites of APP movement and exocytosis. All together, these data identify the exocyst as a key insulin-regulated mediator of neuronal APP trafficking and A{beta} secretion.