Elongasome Dysfunction Triggers Dependence on MepM-Mediated Peptidoglycan Recycling
Elongasome Dysfunction Triggers Dependence on MepM-Mediated Peptidoglycan Recycling
Shamseddine, L.; Janet-Maitre, M.; Chikhi, A. Y.; Kaur, H.; Pellequer, J.-L.; Attree, I.; Job, V.
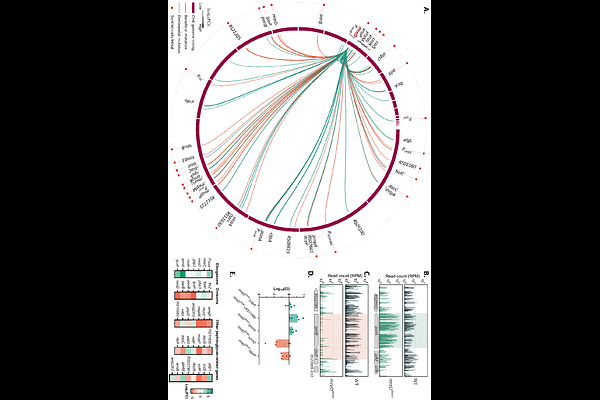
AbstractThe bacterial peptidoglycan (PG) layer is responsible for maintaining cell shape and protecting the cell against both external stress and internal turgor pressure. In rod-shaped bacteria, PG synthesis is orchestrated by two major multi-protein complexes: the elongasome and the divisome, which drive lateral and septal cell wall synthesis, respectively. The membrane protein MreD plays a central role in regulating the elongasome activity. Here, we leveraged a viable Pseudomonas aeruginosa transposon mutant, with reduced mreD expression, to dissect cellular adaptations to impaired elongasome function. A genome-wide synthetic lethality screen revealed that cell division and PG recycling pathways become essential in the context of an impaired elongasome machinery. Moreover, Tn-seq analysis identified the endopeptidase MepM as essential in the mreD downregulated background, whereas its overexpression promoted cell elongation in the parental strain, supporting a conserved role in lateral wall biogenesis. We demonstrate that MepM sustains viability during elongasome impairment by maintaining an active MurU-dependent PG recycling pathway. Together, our findings uncover a functional coupling between PG hydrolysis, recycling, and synthesis. We also identify MepM and PG recycling as critical determinants of bacterial survival when elongasome activity is compromised, positioning these pathways as attractive targets for the development of next-generation or combinatory antibacterial therapies.