APOBEC3 antagonism fully explains HIV-1 Vif essentiality under interferon and differentiation conditions
APOBEC3 antagonism fully explains HIV-1 Vif essentiality under interferon and differentiation conditions
Shimizu, R.; Jonathan, M.; Terasawa, H.; Saito, A.; Monde, K.; Ikeda, T.
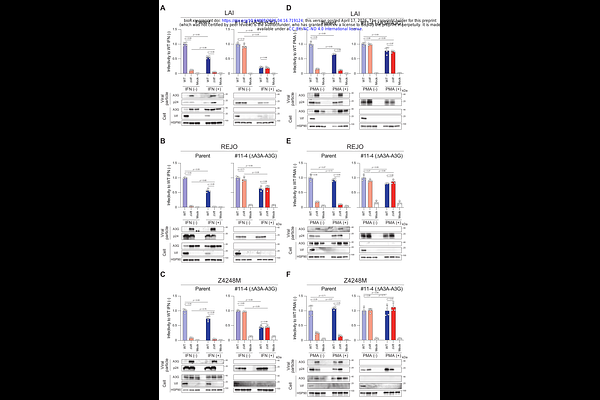
AbstractHIV-1 virion infectivity factor (Vif) counteracts APOBEC3 (A3) proteins by targeting them for proteasomal degradation, thereby preventing lethal G-to-A mutations and loss of infectivity. However, Vif also degrades additional cellular proteins, raising the possibility that its essential role in infectious virion production may extend beyond A3 antagonism, particularly under inflammatory or differentiation conditions. Whether such conditions reveal additional essential Vif targets remains unresolved. Here, using interferon (IFN)-stimulated or phorbol 12-myristate 13-acetate (PMA)-differentiated THP-1 monocytic cells, we directly addressed this question. Although type I IFN and PMA markedly suppressed viral production through Vif-independent mechanisms, {Delta}Vif viruses produced from stimulated parental cells exhibited severely reduced infectivity. In contrast, disruption of A3A-A3G fully restored {Delta}Vif infectivity to wild-type levels under all conditions tested. G-to-A mutations were attributable exclusively to A3 proteins under both IFN and PMA stimulation, whereas IFN-induced, A3-independent blocks to reverse transcription were not antagonized by Vif. Across diverse HIV-1 strains, the requirement for Vif was strictly dependent on A3 family proteins. These findings demonstrate that the essential role of Vif is fully explained by antagonism of A3-mediated restriction and that evolutionary pressure on Vif is driven predominantly by the need to counteract A3-mediated restriction and mutagenesis.