Independent Trafficking and Diverse Stability of XyG-Synthesizing Glycosyltransferases in Distinct Golgi cisternae
Independent Trafficking and Diverse Stability of XyG-Synthesizing Glycosyltransferases in Distinct Golgi cisternae
Zhang, N.; Uthe, K.; Zabotina, O. A.
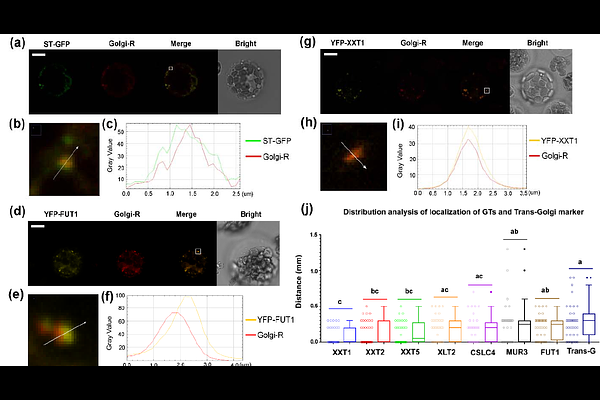
AbstractXyG-synthesizing glycosyltransferases (GTs) are localized in Golgi, and their protein-protein interactions suggest the formation of multiprotein complexes; however, the mechanisms underlying protein complex assembly and transportation, protein stability and degradation remain unknown. We have uncovered that protein-protein interactions among XyG-synthesizing GTs are not prerequisites for Golgi localization and different GTs are first delivered to Golgi as independent proteins. By employing the transient expression of YFP-fused GTs along with a cis-Golgi marker and generating fluorescence intensity profiles, we demonstrated the differential distribution of GTs in Golgi apparatus. We treated Arabidopsis seedlings expressing GTs with cycloheximide (CHX) to estimate half-lives, The GTs exhibit distinct half-lives, and based on their turnover rates, divided into two groups. Our findings revealed that cellulose synthase-like C4 (CSLC4), galactosyltransferase (MUR3), and fucosyltransferase (FUT1) exhibit longer stability. In contrast, XyG xylosyltransferases XXT1, XXT2, XXT5, and galactosyltransferase XLT2 are significantly shorter-living proteins. XyG-synthesizing proteins traffic independently to Golgi, exhibiting distinct sub-Golgi localization that shapes multiprotein complex assembly in specific cisternae. This spatial organization governs partner GT access and residence time for functional efficiency, while GT half-life variations regulate stability and interaction dynamics. Collectively, these factors provide a critical framework for independent operation and coordinated organization of Golgi-resident XyG-synthesizing proteins.