Structural basis of chaperone mechanisms in cells and the evolutionary emergence of the protein world
Structural basis of chaperone mechanisms in cells and the evolutionary emergence of the protein world
Son, A.; Durso, C.; Whitehead, T. A.; Horowitz, S.
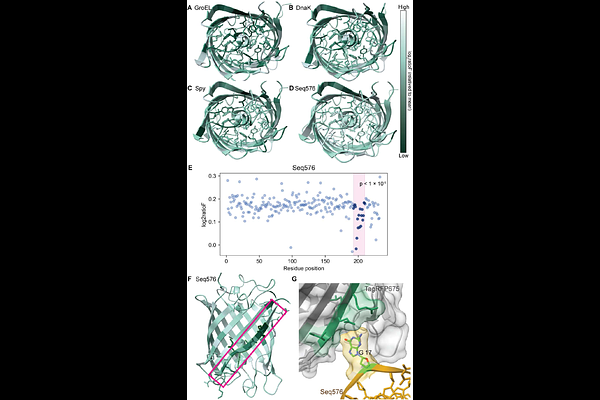
AbstractHow chaperones mediate protein folding in the crowded cell environment remains poorly understood. To gain insight, we developed CHAP-SEQ to examine how chaperones affect protein folding in cells at high throughput and amino acid resolution. Performing CHAP-SEQ using three chaperone proteins and one chaperone RNA reveals distinct modes of folding assistance. Chaperone proteins act preferentially on hydrophobic core residues, whereas chaperone RNA primarily targets structural or dynamic signatures. Furthermore, while the chaperone RNA has little preference for clients' baseline foldability, the chaperone proteins favor clients with greater intrinsic foldability. These differences are consistent with an evolutionary hypothesis in which greater chaperone complexity played a role in the formation of stable hydrophobic cores, suggesting a potential link between chaperone function and the evolution of protein folding.