Dynamic-Structure Redesign of Calmodulin Reveals Mechanistic Constraints on Ryr2 Regulation
Dynamic-Structure Redesign of Calmodulin Reveals Mechanistic Constraints on Ryr2 Regulation
Bogdanov, V.; Tikunova, S.; Fadell, N.; Rebbeck, R. T.; Aprahamian, M. L.; Afsar, M. N. A.; Chekodanov, A.; Blackwell, D. J.; Knollmann, B. C.; Cornea, R. L.; Kekenes-Huskey, P. M.; Lindert, S.; Johnson, C. N.; Gyorke, S.; Davis, J. P.
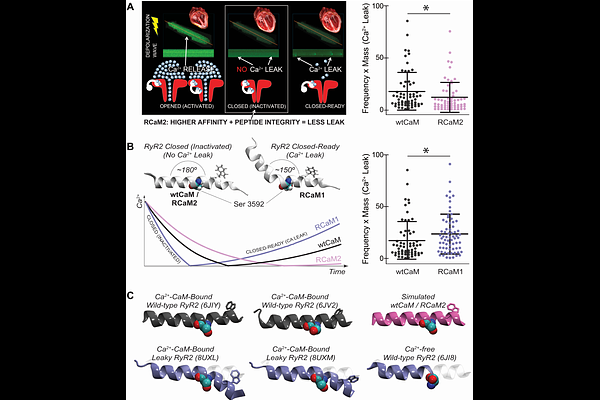
AbstractCalmodulin (CaM) is a highly conserved Ca2+ sensor that regulates hundreds of cellular targets through Ca2+ -dependent conformational dynamics. Despite its central role in Ca2+ signaling and disease, its evolutionary conservation and structural flexibility have suggested that CaM is resistant to rational redesign. Here, using the cardiac Ca2+ release channel Ryanodine receptor 2 (RyR2) as a model system, we tested whether incorporating conformational dynamics into computational protein design enables functional reengineering of CaM. We first applied a static structure-based redesign to increase CaM-RyR2 affinity. Although the resulting variant bound more tightly to both the RyR2 peptide and the intact channel in vitro, it distorted peptide geometry and worsened Ca2+ leak in cardiomyocytes ex vivo. Guided by molecular dynamics simulations, we then developed a dynamic-structure redesign strategy that preserves conformational integrity while strengthening binding. The resulting CaM variant exhibited increased RyR2 affinity and reduced pathological Ca2+ leak in a disease-relevant model. These findings show that improved binding affinity alone is insufficient to enhance physiological regulation and that successful CaM redesign requires preservation of conformational dynamics. More broadly, they demonstrate that integrating conformational dynamics into protein redesign can enable functionally predictive engineering of flexible regulatory protein-protein interactions.