Determinants of CCT motif specificity in WNK signaling and expansion of CCT like domains
Determinants of CCT motif specificity in WNK signaling and expansion of CCT like domains
Magana-Avila, G.; Rojas-Ortega, E.; Lira-Castaneda, M.; Diaz-Ortiz, I.; Bustamante, J.; Carbajal-Contreras, H.; Rojas-Juarez, E.; Ortega-Prado, R.; Marquez-Salinas, A.; Vazquez, N.; Gamba, G.; Castaneda-Bueno, M.
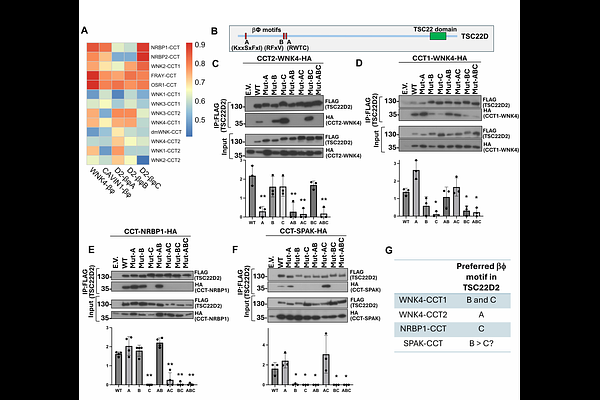
AbstractWNK kinases regulate ion transport and cell volume through interactions with partner proteins such as SPAK, OSR1, NRBP and TSC22D proteins. These interactions are mediated by conserved C-terminal (CCT) domains that recognize short sequence motifs, but the rules governing this recognition remain incompletely defined. Here we show that these domains can be grouped into distinct structural classes with different binding preferences for motif variants. We identify a previously unrecognized motif that mediates binding to the second CCT domain of WNKs and show that interaction specificity is determined by conserved physicochemical features, including electrostatic contacts and aromatic interactions, rather than strict sequence conservation. We further identify a similar domain in the protein FERRY3 that can bind TSC22D motifs in isolation. These findings define a framework for interaction specificity in WNK signaling and suggest that this binding mechanism may extend beyond this pathway.