The Posidonia oceanica Large PSI-LHCII supercomplex reveals the molecular basis of PSI spectral diversification in higher plants
The Posidonia oceanica Large PSI-LHCII supercomplex reveals the molecular basis of PSI spectral diversification in higher plants
Charras-Ferroussier, Q.; Alsenani, T.; Al-Amoudi, A.; Siponen, M. I.; Heilmann, E.; Naschberger, A.; Jungas, C.
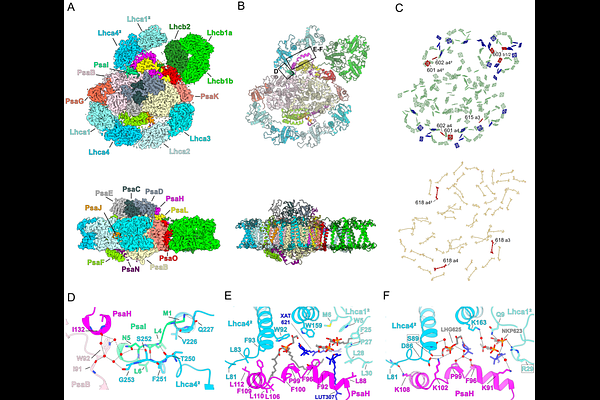
AbstractLand plant Photosystem I (PSI) can capture low-energy photons and convert them into chemical energy through special far-red absorbing chlorophylls, the red-forms. Although the spectral diversity of the red-forms stepped up during land plant evolution, its molecular basis remains largely unknown. The Mediterranean seagrass Posidonia oceanica evolved a large PSI-light-harvesting complex I and II supercomplex (L-PSI-LHCI-LHCII) with blue-shifted absorption and strongly attenuated red-forms. Here, we report the 1.9 A cryo-EM structure of this unique and blue-adapted photosystem, comprising a PSI core with four canonical Lhca proteins (Lhca1-4), a canonically bound LHCII trimer, and an additional Lhca1-Lhca4 dimer anchored through amino-acid residues largely conserved in both marine and land plants. The sub-2 A map revealed several pigment substitutions in the LHC's and further identified amino-acid sites shaping the local red-form environments. Comparison with land plants showed substitution patterns at those sites consistent with the spectral diversification of PSI. These findings suggest a shared structural basis for red-form tuning in higher plants, accounting both seagrass adaptation to seawater light and the broad extent of far-red absorption in land plants.