Repeated mutation of a GT92 glycosyltransferase gene confers antiviral resistance in two Caenorhabditis species
Repeated mutation of a GT92 glycosyltransferase gene confers antiviral resistance in two Caenorhabditis species
Richaud, A.; Zhang, G.; Alkan, C.; Martynow, D.; Belicard, T.; Takeda, N.; Tecle, E.; Felix, M.-A.
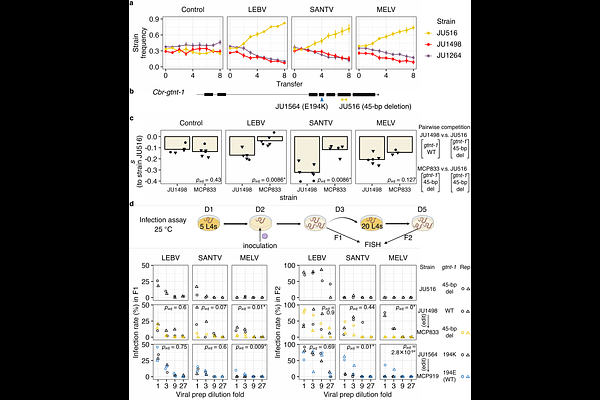
AbstractHost-pathogen interactions evolve rapidly within species, providing natural genetic resources for the identification of specific ecological interaction factors. We previously identified RNA viruses that infect the nematodes C. elegans and C. briggsae in a species-specific manner. Wild strains of both host species demonstrate ample variation in viral sensitivity. Specifically, the wild C. elegans strain MY10, despite carrying a deletion in a key immunity factor, was among the most resistant strains. Here we use recombinant inbred lines and pool-sequencing approaches to genetically map the major MY10 resistance locus, narrowing down its position by CRISPR/Cas9 mediated recombination and testing candidates by genome editing. A rare non-synonymous polymorphism in the gtnt-1 gene, encoding a putative glycosyltransferase of the GT92 family, causes resistance to viral infection in MY10. We find that viral resistance through gtnt-1 mutation occurred repeatedly in C. elegans, with diverse resistance alleles each remaining at low frequency (<1%). Furthermore, leveraging closely related C. briggsae strains differing in viral susceptibility, we demonstrate that repeated reduction-of-function alleles of the Cbr-gtnt-1 ortholog similarly impair viral infection and enhance host fitness upon infection. In conclusion, we found recurrent evolution in two host species of reduction-of-function alleles of the gtnt-1 orthologs, which repeatedly lead to viral resistance yet remain at low frequency. These repeated events provide a case of transient ecological adaptation to a pathogen through recurrent mutation of the same gene in two species. The low population frequencies of the resistant alleles point to a changing eco-evolutionary context that prevents their spread in populations, resulting in high allelic heterogeneity.