Acyl-enzyme dynamics, tautomerisation and hydration regulate turnover of carbapenem antibiotics by the OXA-48 β-lactamase
Acyl-enzyme dynamics, tautomerisation and hydration regulate turnover of carbapenem antibiotics by the OXA-48 β-lactamase
Hoff, J. F.; Beer, M.; Hinchliffe, P.; Tooke, C. L.; Schofield, C. J.; van der Kamp, M. W.; Mulholland, A. J.; Spencer, J.
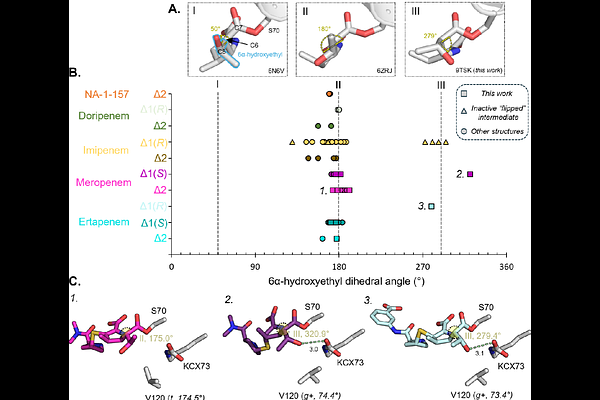
AbstractOXA-48 is a globally disseminated class D serine {beta}-lactamase that efficiently confers resistance to a range of {beta}-lactam antibiotics, including carbapenems, the most potent such agents versus Enterobacterales (Escherichia coli and relatives). Here we characterise the interactions of OXA-48 with the acyl-enzyme complex intermediates formed on its reaction with the carbapenems meropenem and ertapenem using X-ray crystallography and molecular dynamics (MD) simulations. X-ray crystal structures identify acyl-enzymes in both the {Delta}1-imine and {Delta}2-enamine pyrroline tautomeric forms. MD simulations show the epimeric {Delta}2 tautomers of meropenem and ertapenem to more frequently adopt binding poses competent for hydrolysis, i.e. with an appropriate orientation of the carbapenem 6-hydroxyethyl group and positioning of the water molecule required for deacylation; the results indicate that the {Delta}2 tautomers are preferred for deacylation over the {Delta}1-tautomer. MD simulations based on the crystal structures show that, compared to OXA-48, acyl-enzyme complexes of OXA-519 (a natural OXA-48 variant with a single Val120Leu substitution adjacent to the catalytic general base) more frequently sampled conformations favouring hydrolysis, or formation of the alternative {beta}-lactone deacylation product. MD simulations of complexes derived from quantum mechanics/molecular mechanics (QM/MM) simulations show the meropenem-derived {beta}-lactone product is better retained in the OXA-48 active site than hydrolysed meropenem, consistent with reversible {beta}-lactone formation. Overall, our results demonstrate how acyl-enzyme tautomerisation, dynamics and hydration collectively modulate degradation of 1{beta}-methyl carbapenems by class D {beta}-lactamases of the OXA-48 group, and how subtle changes in active site structure potentiate such effects in the OXA-519 variant.