Attenuated interferon signalling in alveolar epithelium limits resistance to Streptococcus pyogenes
Attenuated interferon signalling in alveolar epithelium limits resistance to Streptococcus pyogenes
Wiyana, J.; Turner, D. L.; Amoozadeh, S.; Venkat, P.; Patatsos, K.; Frost, H.; Osowicki, J.; Voss, J.; Chheng, K.; Azzopardi, K.; Caltabiano, N.; Davies, M.; Ramialison, M.; Satzke, C.; Rossello, F.; Steer, A.; Stanley, E.; Werder, R. B.
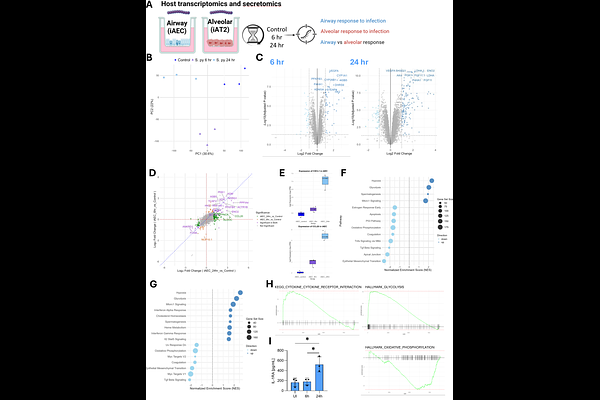
AbstractThe upper respiratory tract is a primary niche for Streptococcus pyogenes colonisation and disease. Lower respiratory tract infection (pneumonia) is the most common invasive S. pyogenes syndrome. Studies have not previously examined how epithelial cells, from the airway to the alveolus, respond to S. pyogenes infection. Here, we established a scalable human in vitro model by differentiating induced pluripotent stem cells (iPSCs) into mature pseudostratified airway epithelium or alveolar type 2 epithelial cells, cultured at air-liquid interface and infected with S. pyogenes (M1UK and M75 strains). Both strains attached to iPSC-derived lung epithelial cells, with significantly greater adherence to the airway epithelium by M75 compared to M1UK. Moreover, invasion by both S. pyogenes strains of alveolar epithelial cells was greater than for the airway epithelium. Dynamic S. pyogenes gene expression changes were evident between 6 and 24 hours after infection, which was influenced by the infected cell type; however, virulence genes were not significantly altered. While infection of the airway epithelium induced rapid and dynamic inflammatory signalling, the alveolar epithelium demonstrated augmented cell death and mounted a transcriptional pro-inflammatory and proliferative response that was uncoupled from cytokine secretion. The airway epithelium model exhibited consistently higher baseline type I interferon (IFN) signalling than the alveolar epithelium. Invasion by S. pyogenes and inflammation was significantly reduced in IFN-{beta}-treated alveolar epithelial cells. In summary, we have established the first model of S. pyogenes infection in physiologically relevant airway and alveolar epithelial cells. Our findings suggest that host responses to infection are influenced by lung compartment, the S. pyogenes strain type, and infection timepoint, highlighting context-specific pathways that could be leveraged therapeutically.