Topographically segregated mediodorsal-prefrontal loops exhibit distinct cue dynamics during learning and extinction
Topographically segregated mediodorsal-prefrontal loops exhibit distinct cue dynamics during learning and extinction
Runyon, K.; Sanders, K.; Hartle, A.; Howe, W. M.
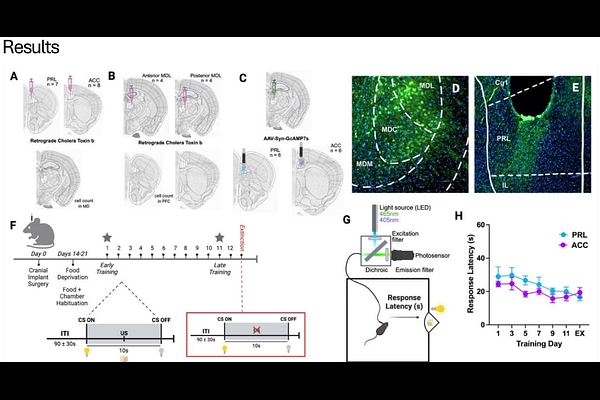
AbstractUsing external cues to guide behavior is a core function that enables multiple aspects of cognition and attentional control, and deficits in this process are central to many theories of neuro-psychiatric, degenerative, and developmental disorders. Cue detection relies on the precise coordination of neural circuits, with the mediodorsal thalamus (MD) hypothesized to play a pivotal role in orchestrating the relay of cue-based associative information to the prefrontal cortex. The prefrontal cortex comprises multiple subregions, which are believed to differentially contribute to such associative cue-based behaviors. This regional specificity is likely seeded by projection-defined MD[->]PFC pathways, although the anatomical organization of these discrete channels and their dynamic roles in cue detection are still being defined. Here, we address this gap by combining anatomical circuit mapping of MD-PFC output pathways with in vivo calcium imaging during a cue-based reward conditioning task in mice. These experiments reveal that MD projections to distinct PFC subregions (prelimbic and anterior cingulate cortex) form topographically defined loops, that are characterized by unique patterns of activity across cue-reward learning. Using fiber photometry to monitor changes in calcium activity in axonal projections from the MD to the PFC, we show that during learning, MD projections to the prelimbic subregion are activated by cue presentation, and the dynamics of this activity remain stable across training days. In contrast, MD projections to the anterior cingulate exhibit a learning-dependent suppression of activity that predicts reward approach behavior in late training. Interestingly, the two pathways exhibit opposing activity patterns when the predictive validity of the cue is diminished by extinction training, suggesting distinct functional roles in detecting violations of learned contingencies. Together, these findings reveal previously unrecognized anatomical and functional distinctions within MD-PFC circuits and demonstrate that parallel thalamocortical pathways differentially support cue detection and behavioral flexibility. This work advances understanding of thalamocortical mechanisms underlying cue detection and may inform circuit-based approaches for treating cognitive dysfunction in psychiatric disorders.