ARMH3 acts as a central scaffold at the Golgi/TGN through interactions with Arl5, GBF1, and PI4KB
ARMH3 acts as a central scaffold at the Golgi/TGN through interactions with Arl5, GBF1, and PI4KB
Scott, M. K.; Klynsoon, G. C.; Walsh, E. E.; Suresh, S.; Nyvall, H. G.; Burke, J. E.
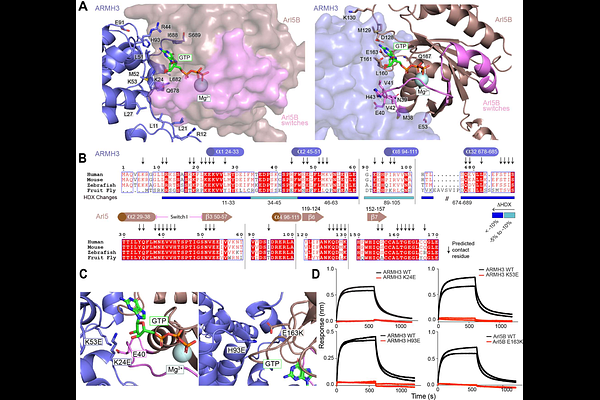
AbstractThe armadillo repeat protein ARMH3 regulates the activity and localisation of the Golgi resident lipid kinase phosphatidylinositol 4 kinase III{beta} (PI4KB) and the Golgi-specific brefeldin A-resistance guanine nucleotide exchange factor 1 (GBF1) that activates Arf1. ARMH3 localises to the trans Golgi network (TGN) via the GTPase Arl5. We used hydrogen deuterium exchange mass spectrometry (HDX-MS) and AI-enabled modeling to define the interfaces of ARMH3 with its binding partners Arl5, PI4KB, and GBF1. The ARMH3-Arl5 interface was determined to consist of the N and C termini of ARMH3, with Arl5 binding causing conformational changes in ARMH3 located at a shared PI4KB/GBF1 interface. Both PI4KB and GBF1 form mutually exclusive complexes with ARMH3, with GBF1 binding to ARMH3 through a disordered loop we have named the ARMH3 binding region (ABR). The ARMH3 interfaces in PI4KB and GBF1 contain phosphosites, with the phosphomimetic mutation of GBF1 blocking complex formation. These findings provide new insights into the role of ARMH3 as a master coordinator of GTPase and phosphoinositide signaling at the Golgi/TGN.