Ratiometric Quantification of Dissolved Molecular Oxygen in Microplates for Biochemical Assays Using Palladium Porphyrin Photoluminescence
Ratiometric Quantification of Dissolved Molecular Oxygen in Microplates for Biochemical Assays Using Palladium Porphyrin Photoluminescence
Podolskiy, D.; Plieth, C.
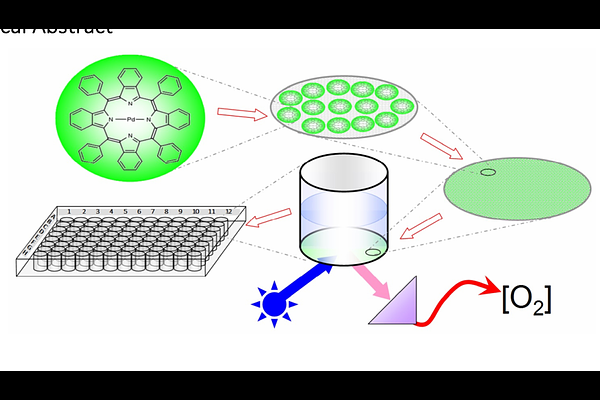
AbstractMany biochemical processes are dependent on the presence or absence of molecular oxygen (O2). Palladium-tetrapyrrol derivatives can be used to measure O2-concentrations and O2-turnover during biochemical reactions and microbial growth in standard microtiter plates (MTPs). Palladium(II)-5,10,15,20-(tetrapentafluorophenyl)-porphyrin (1; CAS 72076-09-6) and Palladium(II)-5,10,15,20-(tetraphenyl)tetrabenzoporphyrin (2; CAS 119654-64-7) are introduced with this study. Spectral analyses of both compounds revealed that fluorescence quenching by O2 is not evenly distributed throughout all wavelengths and can therefore be used ratiometrically. Experimentally determined fluorescence lifetimes are around 500 s and 300 s for 1 and 2, respectively. A simple protocol is disclosed, how to immobilize the indicators on the bottom of MTP wells to give clear transparent dye doped polymer layers. We propose a straightforward procedure of how fluorescence data can be processed and calibrated in terms of O2 concentrations. Diverse applications are demonstrated and discussed, which include oxygen consumption and production by microorganisms as well as by enzymatically catalysed biochemical reactions. Various aspects are critically considered, as there are e.g. the dependence of O2 solubility on temperature and salinity, the diffusion of O2 across diverse phase boundaries, the unwanted O2 ingress into the reaction volume, the oxygen binding capacity of the MTP plastic material and the pH-dependence of the sensor layer. The findings and methods presented here open up a broad variety of high throughput assays involving changes of dissolved O2 as measurands for biochemical and biological activity.