Cooperative Control of Arrestin Activation By Membrane Lipids And Phosphorylation Barcodes
Cooperative Control of Arrestin Activation By Membrane Lipids And Phosphorylation Barcodes
Aydin, Y.; Zhuo, Y.; Yen, Y.-C.; Chen, C.-L.; Klug, C. S.; Marchese, A.; Chen, Q.
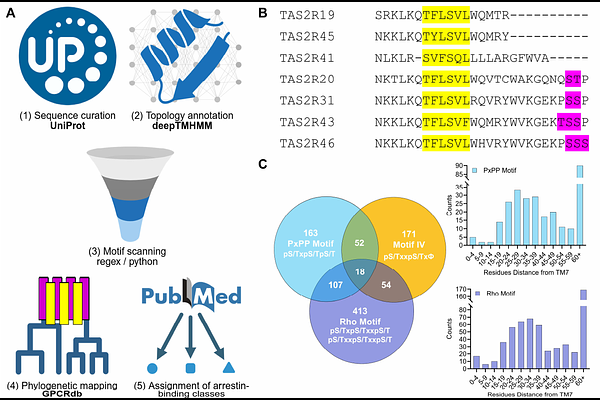
AbstractArrestins regulate G protein-coupled receptor (GPCR) signaling by binding phosphorylated receptors embedded in lipid bilayers, yet how receptor phosphorylation and membrane composition cooperate to control arrestin activation remains unclear. Here, we reconstitute this interplay using N-terminally palmitoylated phosphopeptides tethered to nanodiscs of defined lipid composition and quantitatively measure arrestin-2 (Arr2) activation and membrane engagement. We find that both receptor phosphorylation and the lipid environment are essential for robust Arr2 activation, with phosphoinositides (PIPs) and other anionic lipids facilitating Arr2 activation and membrane association through distinct mechanisms. Systematic profiling of phosphorylation barcodes derived from atypical chemokine receptor 3 (ACKR3) and vasopressin receptor 2 (V2R) identifies phospho-motifs that potently activate Arr2. Moreover, the position of these motifs relative to the membrane determines Arr2 engagement, supporting a model of regional phosphorylation barcodes. Genome-wide motif analysis further links the phosphorylation barcode to predicted arrestin coupling strength and classification into Class A or Class B GPCRs. Finally, lipidated phosphopeptides inhibit GPCR-Arr2 interactions in live cells and enable structural characterization of Arr2-phosphopeptide complexes by cryo-electron microscopy, establishing a membrane-integrated framework for decoding arrestin response.