Architecture-Dependent Stability, Cellular Uptake, and Redox Modulation of Poly(p-Coumaric Acid) Hybrid Nanoparticles for Ovarian Carcinoma Intervention
Architecture-Dependent Stability, Cellular Uptake, and Redox Modulation of Poly(p-Coumaric Acid) Hybrid Nanoparticles for Ovarian Carcinoma Intervention
Megahed, M.; Harun, A.; Herrera, K.; Rashid, M. H.; De Leon, I.; Srivastava, I.
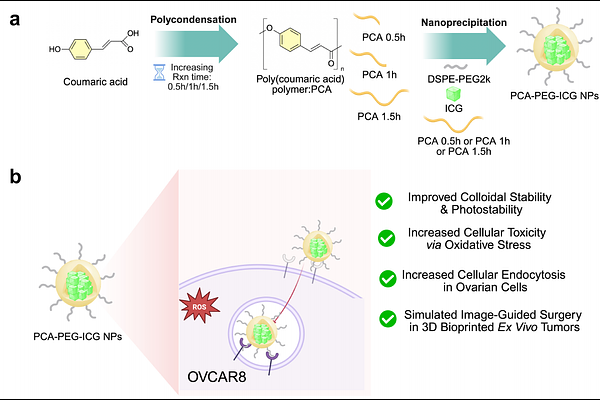
AbstractThe clinical efficacy of fluorescence-guided surgery (FGS) is often compromised by the poor photostability and biologically inert nature of conventional contrast agents like Indocyanine Green (ICG). While nanocarriers can enhance dye stability, they typically function as passive delivery vehicles, requiring additive complexity to achieve therapeutic effects. Here, we report a structure-guided approach to develop self-theranostic hybrid nanoparticles where the polycondensation kinetics of the polymer core, poly(p-coumaric acid) (PCA), serve as a master switch for both optical and biological performance. By systematically tuning the reaction duration, we synthesized PCA variants with distinct macromolecular growth profiles that dictate the efficiency of nanoparticle assembly and hydrophobic sequestration. Optimized PCA architectures significantly enhanced the photostability of encapsulated ICG via nanoprecipitation, maintaining robust NIR-I fluorescence under continuous surgical illumination. Beyond acting as a structural scaffold, the PCA matrix retains intrinsic redox activity, triggering apoptosis in ovarian cancer cells via the modulation of reactive oxygen species (ROS) generation. The translational utility of these nanoparticles was validated using 3D bioprinted intraperitoneal tumor phantoms, which recapitulate the optical scattering and spatial heterogeneity of metastatic disease to enable high-contrast surgical simulations. This work establishes reaction-time-dependent architecture as a critical design parameter for integrating imaging and intrinsic therapeutic functionalities within a single, simplified nanomaterial platform.