Native entanglement misfolding contributes to age-associated structural changes across the Saccharomyces cerevisiae proteome
Native entanglement misfolding contributes to age-associated structural changes across the Saccharomyces cerevisiae proteome
Vu, Q. V.; Sitarik, I.; Nissley, D. A.; O'Brien, E. P.
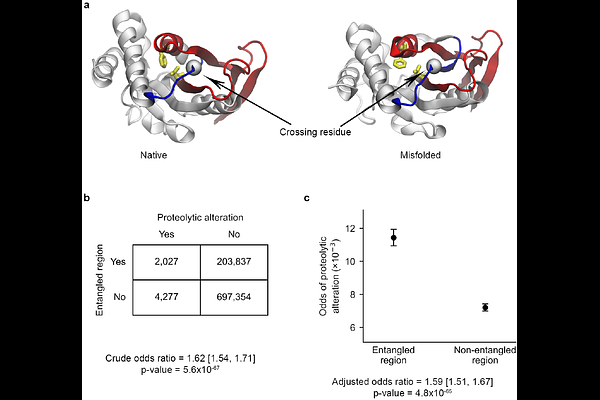
AbstractAging at the subcellular level involves the simultaneous decline in the cell's ability to maintain protein homeostasis and rise in misfolded proteins through a positive feedback loop. Here, we test if a widespread class of protein misfolding could contribute to proteome aging by examining if statistical associations exist between age-related changes in protein structure, measured by limited proteolysis mass spectrometry data of the aging Saccharomyces cerevisiae proteome, with structural annotations and molecular simulations. We find that globular proteins that are likely to exhibit entanglement misfolding are 121% more likely to exhibit age-related structural changes, and these changes are 59% more likely to be localized to natively entangled regions. Proteins containing native entanglements are seven-fold more likely to misfold, according to simulations, and populate long-lived, near-native misfolded states. Thus, the age-related structural changes in yeast proteins can be explained in part by the accumulation of misfolded proteins involving entanglements.