MRI-Based Blood Clot Phenotyping: An In Vitro Study
MRI-Based Blood Clot Phenotyping: An In Vitro Study
Bechtel, G. N.; Das, A.; Noyer, J.; Bush, A. M.; Hormuth, D.; Yankeelov, T. E.; Castillo, E.; Warach, S.; Fuhg, J.; Tamir, J. I.; Saber, H.; Rausch, M. K.
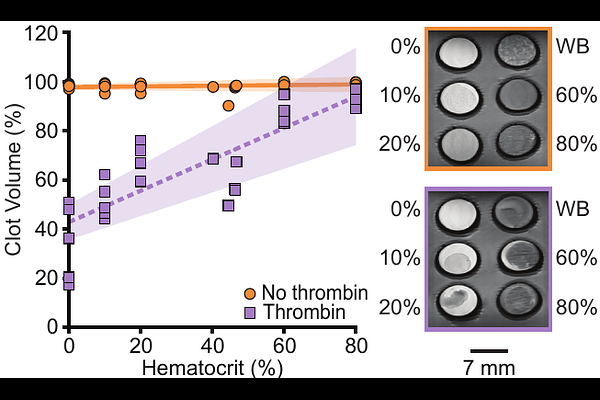
AbstractBackground and Purpose: Neurointerventional outcomes depend on clot composition and may be influenced by clot contraction. Thus, a priori identification of clot composition and contraction could inform procedural strategies and improve outcomes. The goal of our work is to conduct an in vitro test to determine whether MRI can reliably predict both clot composition and contractile state. Materials and Methods: To this end, we prepared blood clots spanning clinically observed compositions (0-80% red blood cells (RBCs)) in both contracted and uncontracted states. Contraction was controlled by coagulating blood with or without thrombin. We imaged these clots using quantitative, clinical, and investigational MRI sequences. Using these data, we then determined whether MRI signal intensities, quantitative parameters, and radiomic features capturing intensity and texture patterns can (i) predict clot hematocrit and (ii) classify clots by composition (RBC-rich vs. fibrin-rich) and contraction state. Results: Quantitative MRI parameters (T1, T2, ADC) decreased with increasing hematocrit (R2 = 0.56-0.85, p < 0.001), while signal intensities from clinical sequences showed weaker correlations (R2 = 0.46-0.62, p < 0.001). Radiomic models predicted hematocrit with performance comparable to MRI parameters. When applied to classification, radiomic features accurately discriminated RBC- versus fibrin-rich clots, with AUCs exceeding 0.90 across nearly all sequences. In contrast, classification of contraction state showed greater variability in AUCs across sequences but remained high for quantitative T1 and T2 values (AUCs up to 0.88). Trends were consistent across clots coagulated with and without thrombin. Pooling features across sequences did not outperform the best individual sequence for either regression or classification. Conclusions: We demonstrate that MRI-based radiomic analysis quantitatively characterizes clot composition and contraction in vitro. These findings support the feasibility of using MRI for pre-interventional clot phenotyping, with potential to inform thrombolytic and mechanical thrombectomy strategies. Thus, in vivo studies validating these results are warranted.