Histone H1 Variants Regulate Neurodevelopmental Transcriptional Programs in Autism with 16p11.2 deletion
Histone H1 Variants Regulate Neurodevelopmental Transcriptional Programs in Autism with 16p11.2 deletion
Brudno, R.; Askayo, D.; Khair, D.; Shayevitch, R.; Keydar, I.; Zmudjak-Olevson, M.; Lev-Maor, G.; Zavolan, M.; Elkon, R.; Ast, G.
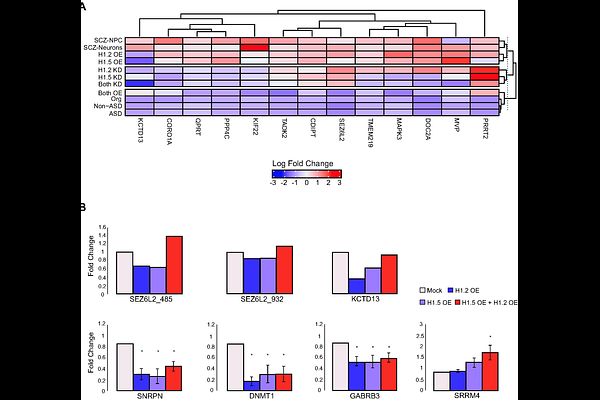
AbstractBackground: Neurodevelopmental disorders, including autism spectrum disorder, involve widespread transcriptional dysregulation. Copy number variations at 16p11.2 are among the strongest genetic risk factors for autism spectrum disorder, yet the molecular mechanisms by which these copy number variations contribute to neurodevelopmental pathology remain unclear. Results: We identify significant genetic associations between autism spectrum disorder susceptibility and the HIST1 histone gene cluster through genome-wide analysis. Transcriptomic profiling across post-mortem brain tissue, patient-derived neural progenitor cells, neurons, and cerebral organoids reveals consistent upregulation of linker histone variants H1.2 and H1.5 in idiopathic autism spectrum disorder and 16p11.2 hemi-deletion carriers, but not in schizophrenia or bipolar disorder. Functional assays demonstrate that dysregulated H1 expression disrupts gene networks involved in synaptic signaling, chromatin remodeling, and neural differentiation. Mechanistically, we link H1 upregulation to MAZ, a transcription factor encoded within the 16p11.2 locus. MAZ binds the promoter regions of H1 genes and represses their transcription. Knockdown of MAZ leads to H1 overexpression. H1 upregulation alone is sufficient to alter the expression of autism spectrum disorder-associated genes. Conclusions: Our findings define a MAZ-dependent regulation of H1 dosage as a critical chromatin-mediated mechanism contributing to transcriptional pathology in 16p11.2-associated autism spectrum disorder. Keywords: Histone H1, Autism Spectrum Disorder, 16p11.2 Hemi-deletion, MAZ, chromatin remodeling, transcriptomics.